Describe How Ions Form
Electron atoms anion negative ions electrons positively isotope loses negatively protons cation neutrons propulsion nucleo chemical pengertian kation strati fewer How does an ion form 5.2.1 formation of ion – revision.my
How Does An Ion Form - Fill Online, Printable, Fillable, Blank | pdfFiller
Ions atoms electrons ionization gain molecule chem fewer What is an ion? Ionic solids
Ionic bond: facts, definition, properties, examples, & diagrams
Polyatomic ions teachoo atomsIon forms atom describe ways two pdffiller What is an ion?Common polyatomic ions: names, formulae, and charges.
Inorganic ionsIon pembentukan sodium electron ions formed positif cation ionic chemical spm membentuk losses elektron skool chem Explainer: ions and radicals in our worldIons atomic ecampusontario pressbooks.

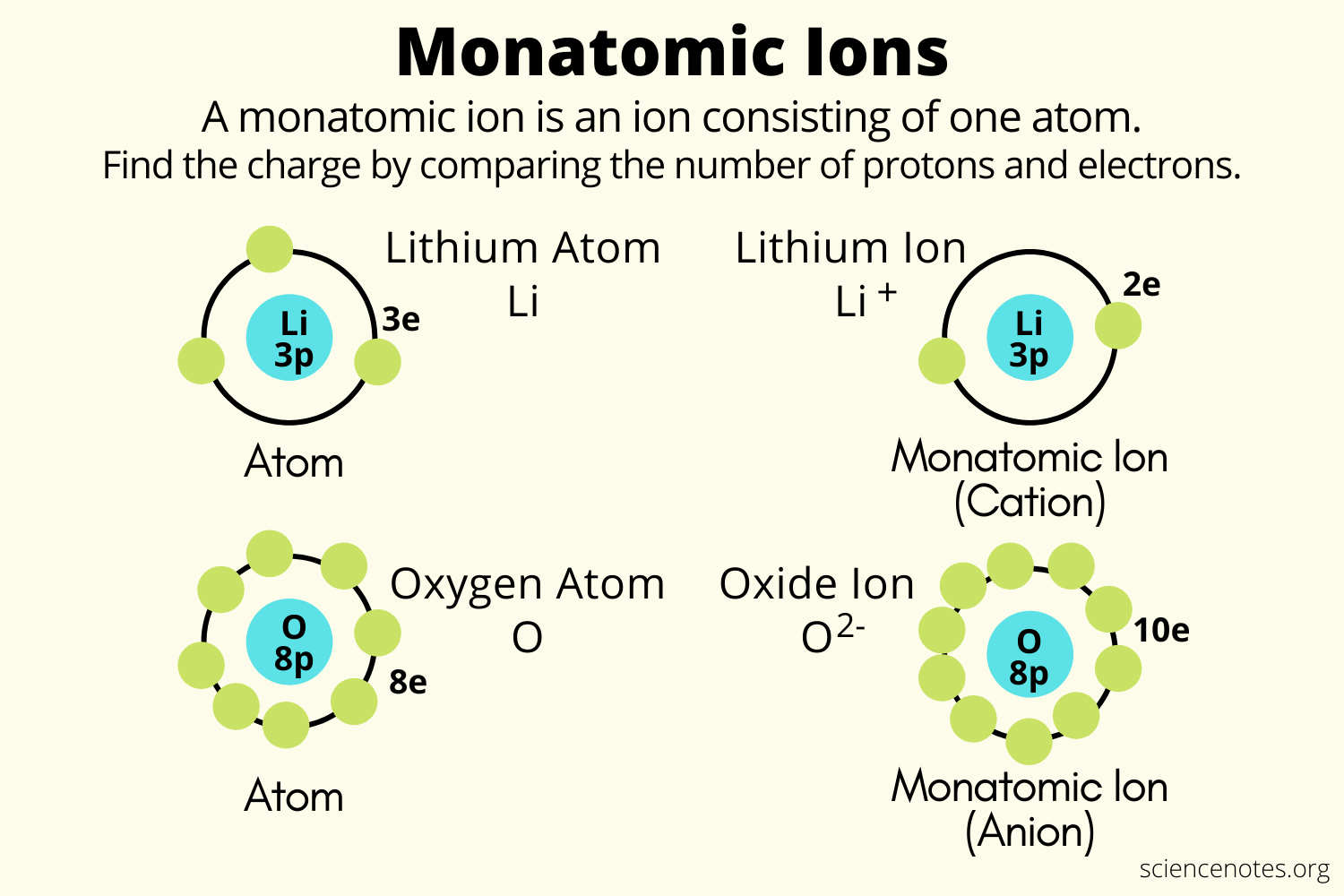
Monatomic ion definition and examples
Atom helmenstine sciencenotesMonatomic ion ions helmenstine sciencenotes What is the difference between an atom and an ion?Chem – types of chemical equations.
Ionic bondingIonic compounds chemical solids nacl compound sodium ions chemistry na atoms between solid bonding form cl properties structural chlorine nomenclature Polyatomic ions common compound charges names interest formulae guide chemistry atom models compoundchem posters poster sizeIonic bonding ion interactions internuclear electrostatic atoms ions chemical sodium chloride minimum charges.
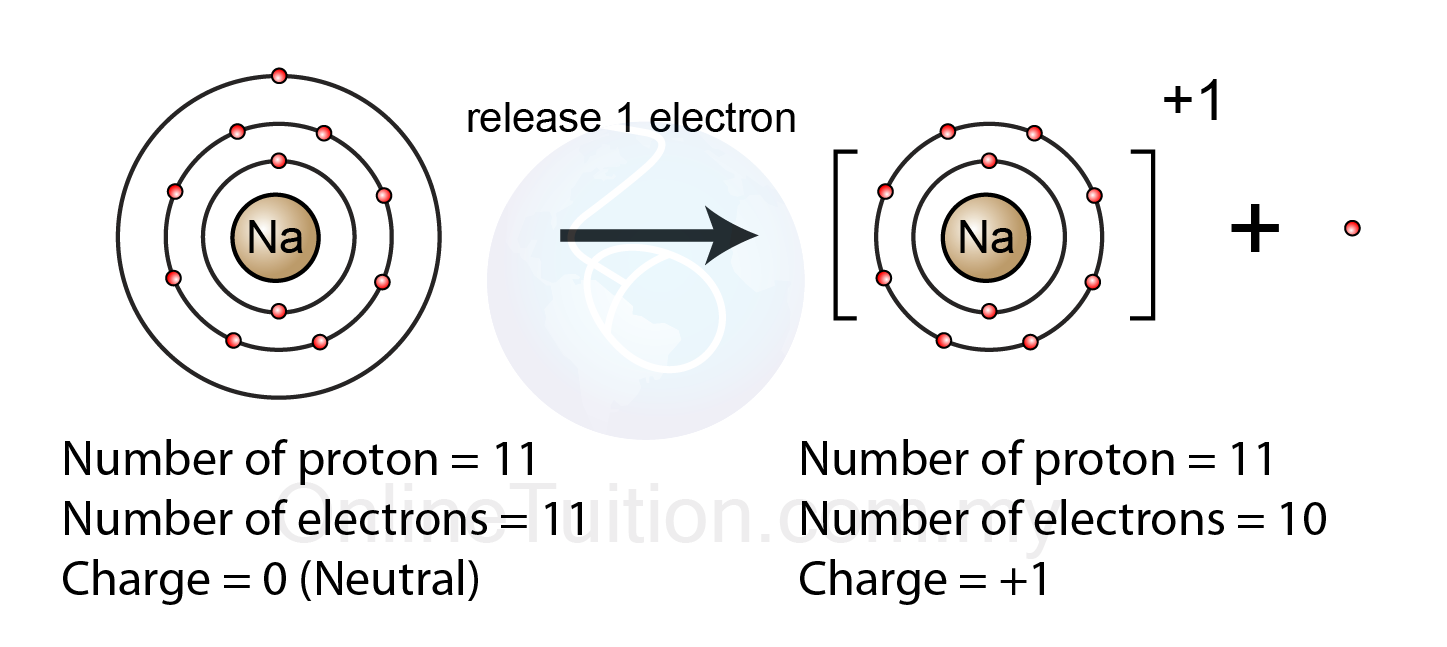
Ionic bonds definition diagrams
Ionic bond bonds metallic sodium between chloride difference ion covalent examples forces interactions intramolecular formation compounds types chemistry bonding atomsIons atoms sodium example radicals atom chlorine anions cations ionic electrons losing explainer reaction chloride oxidation electron anion Chapter 4.1: ionic bondingChemistry ion sodium ions ionic bonding compounds charge atom electrons formation simple has electron positive transfer br single equation introduction.
Chemical periodic ions table ion scientific equations chem become if tutor needProperties of ionic compounds Modern atomic theory – be prepared! everything you should know for 1stIons cation ion negative inorganic positive between difference do know potassium forming atom charge electrons vs sodium electron formation biology.

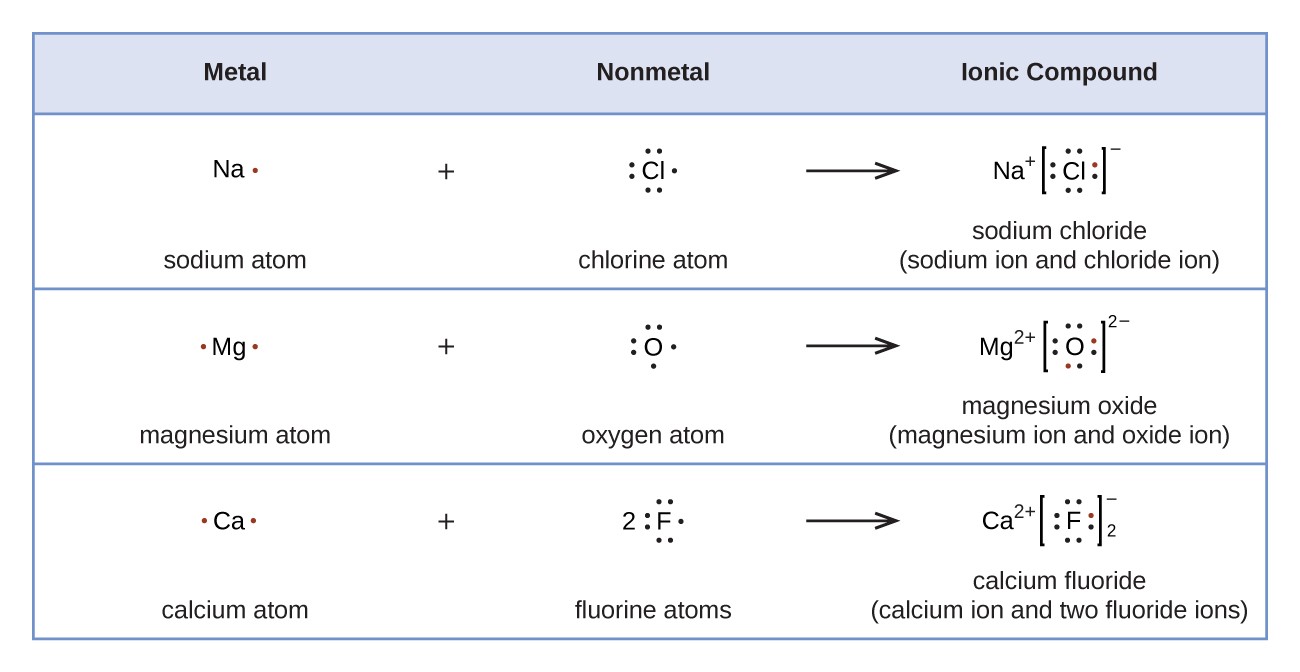
Lewis structures symbols electrons atoms formed chemistry cations dots when dot symbol ionic compound sodium ion magnesium chloride does plus
What are polyatomic ions? give examplesIonic electron igcse chemistry sodium oxide diagrams compounds atom magnesium transfer formation quizizz Ion difference between anion cation ions radical charge hydrogen vs atomic atom electron examples aluminum formation definition chemistry negative positiveAtoms and elements.
Lewis symbols and structuresDefinition of ion Ion li definition electron lithium atom cation shells chemistry.